Board Highlights - June 14, 2019
Highlights from this Board Meeting include a number of PODSA modernization amendments, data from PRP’s Registrant Feedback Survey; and an update on the College’s organizational efforts with regards to Excellence Canada Certification and its next Strategic Plan.
June #CPBCBoard meeting (morning) https://t.co/BsCTaF8FjV
— BC Pharmacists (@BCPharmacists) June 14, 2019
Practice Review Data Report & Registrant Feedback Survey Report
Vice-Chair of the Practice Review Committee, Mike Ortynsky and Community Pharmacy Compliance Officer, James Van presented to the Board the data and practice insights gained from pharmacy and pharmacy professional reviews as well as from the registrant feedback survey.
The Practice Review Program (PRP) is a comprehensive cyclical review of pharmacies and pharmacy professionals completed to ensure the standards of the College of Pharmacists of British Columbia are met. The reviews are conducted around focus areas identified by the Board as having the greatest impact on patient safety including patient identification verification, profile check, counselling, documentation, product distribution and collaboration.
The Practice Review Program is committed to being receptive and responsive through the continual monitoring and evaluation of data and feedback, and implementing iterative changes and improvements to the program based on that data and feedback.
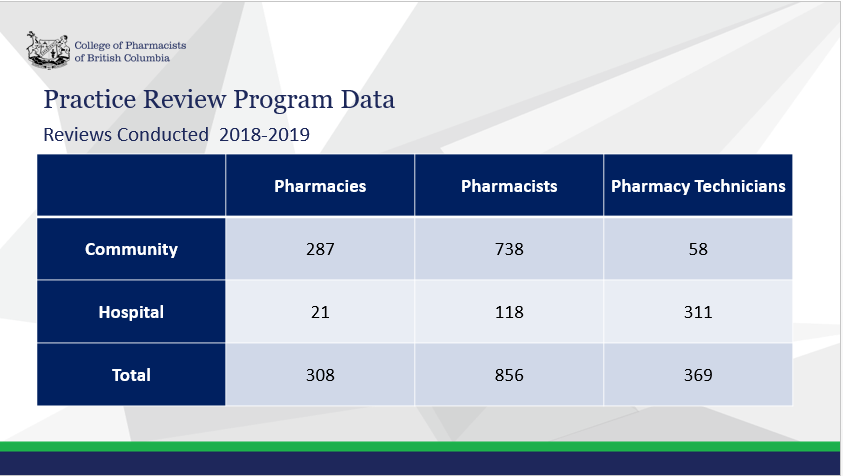
While the results have been generally positive, the PRP department recognizes that more work needs to be done to further improve compliance and positively impact patient safety.
Throughout the practice review process, areas of non-compliance are identified, documented, and resolved. For program evaluation, development, and education purposes, the 5 most frequent non-compliance categories for each area reviewed are identified.
Based on data collected during reviews, the following top areas of non-compliance were identified:
Top areas of non-compliance in Community Pharmacy:
- Security
- Equipment and References
- Prescriptions
- Inventory Management
- Pharmacy Manager Responsibilities
Top areas of non-compliance in Hospital Pharmacy:
- Sterile Compounding
- Inventory Management – Nursing Unit
- Pharmacy Manager’s Responsibilities
- Ambulatory Services
- Equipment and References
Top areas of non-compliance in Pharmacist reviews:
Community Practice
- Counselling
- Documentation
Hospital Practice
- Patient Identification Verification
- Counselling
Top areas of non-compliance in Pharmacy Technician reviews:
Community Practice
- Collaboration
- Documentation
Hospital Practice
- Patient Identification Verification
- Documentation
As a result of the collaborative nature of the Practice Review Program, with one-on-one follow up between COs and registrants after reviews, all action items identified were resolved, and there were no referrals to the Inquiry Committee in the 2018-2019 fiscal years.
Registrant Feedback Survey
31% of community, 25% of hospital pharmacy professionals responded to feedback survey. #CPBCBoard pic.twitter.com/0JNJvPeGPY
— BC Pharmacists (@BCPharmacists) June 14, 2019
The survey assessed registrants’ feedback on the review process in a number of areas including PRP tools, the review process itself and the Compliance Officers.
Overall feedback was very positive, with agreement ratings all falling between 80-100%.
A number of changes have been made to the practice review program as a direct result of the feedback received, including:
- No reviews scheduled during peak holiday times
- Increased professional reviews per day where possible
- Increased review timing flexibility
- New pre-review tool IT updates and extension policies
- Added pharmacy technician, residential care and compounding specific questions
- Confirmation phone calls
- Survey design changes
- One business day response time
- Exploring browser compatibility issue
- Program evaluation and ongoing development
Developing the College’s Next Strategic Plan: Goals and Objectives
The Board approved, in principle, draft goals and objectives for the College’s next Strategic plan which will launch in 2020. The Board also approved a proposal for term of the upcoming Strategic Plan to be 2020/21 to 2024/25, permitting five years to complete the action items.
Mary O'Callaghan, sharing college's strategic planning model w/ #CPBCBoard pic.twitter.com/tPwBn95vce
— BC Pharmacists (@BCPharmacists) June 14, 2019
Much of the discussion and considerations into developing draft goals and objectives for theCollege’s next Strategic Plan has focused on the report by Harry Cayton, 'An Inquiry into the performance of the College of Dental Surgeons of British Columbia and the Health Professions Act' (‘the Cayton Report’).
The report outlines an inquiry into the College of Dental Surgeons of British Columbia and recommends changes to the Health Professions Act. As part of considering the Cayton Report, and particular attention was paid to the concepts of “patient safety,” “public health and wellness” and “right touch regulation.” These concepts, align with the College’s mandate and approach to its work.
The College also considered the results of an environmental scan related to emerging trends in pharmacy practice and regulation, as well as input provided through the College’s Strategic Plan Engagement, which received over 7000 comments through an online survey.
Additional topics of interest from strategic planning sessions include:
- Pharmacy care that is: ethical, equitable, inclusive, socially just, respectful, safe and culturally safe.
- Commitment to cultural humility as a path to cultural safety.
- Provision of care and services that respects economic status and social justice.
- People feeling respected (not judged) by pharmacy care professionals.
- Inclusive care: inclusive of patient, family, health care team, and the public.Workload that enables safe practices, such as:
- Safe use of medication: providing education, information, and communication to end user to ensure safe use.
- Technical safety: safe dispensing practices that minimize risk of medication errors.
- Emphasis on evidence-based, patient-centred, and interdisciplinary care.
- “Pharmacy Care Provider” in place of “Pharmacy Professional” to avoid perception of power imbalance inherent in “Professional”.
As a result of these strategic planning sessions, the Committee of the Whole developed the following 4 strategic goals and objectives, in principle:
Strategic Goal One
The public is given evidence-informed, patient-centred, interdisciplinary care.
Objective 1.1: To develop a plan to support the provision of evidence-informed, patient-centred, interdisciplinary care that includes cultural safety and humility.
Strategic Goal Two
To enable practice innovation through regulation that enhances health and wellness of the public and ensures patient safety.
Objective 2.1: Ensure patient safety and health and wellness of the public by implementing a transparent plan that engages the public in identifying practice innovations and determining the College’s role.
Strategic Goal Three
To have the public and health professionals see pharmacy professionals as valuable resources who are acting first and foremost in the public interest.
Objective 3.1: To communicate what the public and health care professionals can expect from pharmacy professionals.
Strategic Goal Four
To have strong, collaborative engagement with all healthcare providers to advance patient-centred, interdisciplinary care.
Objective 4.1 Enhance patient health and wellness by creating an interdisciplinary framework for collaborative engagement with all healthcare providers.
With the approval of the draft Strategic Plan goals and objective, College staff will now begin developing and costing the Strategic Plan to bring back for a final decision at the September Board meeting.
Excellence Canada Update
The College has been awarded Silver Certification with Excellence Canada’s ‘Excellence, Innovation and Wellness Standard.
Key outcomes of EIW Silver - wide understanding by employees of organization's approach to excellence, innovation and wellness, strategic/operational plans in place, establishment of baseline indicators, measures/related goals for EIW. #CPBCBoard pic.twitter.com/c0AOooYBvU
— BC Pharmacists (@BCPharmacists) June 14, 2019
The Excellence, Innovation and Wellness Standard is an integrated quality-based management system, based on a holistic strategic framework that ensures organizations achieve the best possible outcomes across all business drivers, including: Leadership, Planning, Customers, People and Processes. Each standard has four levels of certification (Bronze, Silver, Gold, Platinum), comprised of requirements that facilitate progressive implementation.
This certification helps the College achieve its current strategic goal of Organizational Excellence, which states that:
“…the College will ensure that the efficiency and effectiveness of its foundational business processes and technological supports are upgraded to meet the ongoing needs of registrants, pharmacy owners and directors, staff and the public. It will also ensure that College governance and staffing are well organized and provided at the appropriate level to ensure the efficient and effective delivery of services to all stakeholders.”
In order to approach this goal in an organized fashion, the College partnered with Excellence Canada, who recommended we work towards a Silver Certification in 2019.
The key outcomes of the Silver Certification are:
- Enhanced employee involvement in planning and improvement initiatives
- A wider understanding by employees of the organization’s strategic approach to excellence, innovation and wellness
- Strategic and annual operating plans are in place
- Establishment of baseline indicators, measures and related goals for excellence, innovation and wellness.
College Staff were strongly engaged in this process. Action Teams drafted new policies and/or procedures, presented them to the Excellence Council, the Executive and/or Management Teams and then to full Staff Meetings prior to launching the new policy or procedure.
Additionally, in order to meet the Silver Certification requirements, the Excellence Council had to ensure that all staff were aware of and used the newly created policies and procedures. This was accomplished through staff meeting presentations, staff Intranet articles, as well as a ‘Completion Gauge” graphic on the staff Intranet.
About Excellence Canada
Excellence Canada was originally founded in 1992 by Industry Canada as the National Quality Institute (NQI). In 2011, they were rebranded as Excellence Canada, an independent, not-for-profit Corporation that is dedicated to advancing organizational performance across Canada.
As Canada’s national authority on Quality and Healthy Workplace practices, Excellence Canada has created a uniquely Canadian model, providing measurable standards and objective validation through its certification program.
PODSA Modernization Phase Two Bylaw Amendments
The Board approved for public posting, amendments to the bylaws under the Pharmacy Operations and Drug Scheduling Act (PODSA), regarding the College’s PODSA Modernization initiative.
#CPBCBoard approve PODSA modernization bylaw amendments for public posting. Stay tuned for more information about this public posting! pic.twitter.com/0cIbaHa4xs
— BC Pharmacists (@BCPharmacists) June 14, 2019
As part of our 2017-2019 Strategic Plan, the College conducted a comprehensive review and reform of bylaws and policies established under PODSA.
The College’s review of included ensuring:
- Bylaws are clearer and duplication in bylaws and policies is addressed.
- Professional Practice Policies (“PPPs”) are standardized and transitioned to bylaw where needed.
- Bylaws and PPPs have consistent writing style and structure.
The following key topics were identified from registrant and stakeholder feedback as well as Practice Review Program (“PRP”) data:
High Priority Topics
- Operation of a Community Pharmacy without a Full Pharmacist present.
- Responsibilities of Managers, Direct Owners, Directors, Officers and Shareholders.
- Storage of drugs and confidential health information, including offsite storage.
- House-keeping amendments, including ensuring consistency of writing style.
Medium Priority Topics
- Developing provisions to allow for community telepharmacy reinstatement.
- Determining if certain provisions are better placed in the Health Professions Act Bylaws.
- Reviewing the “Top 10” requirements not being complied with (based on PRP data). The aim of reviewing those requirements is to determine if any bylaw amendments are needed or if other tools (e.g., education, etc.) would assist with enhancing compliance.
- Reviewing PharmaNet requirements in light of the transition of administration of PharmaNet functions to the Ministry of Health.
The amendments will now be publicly posted for comment for a period of 90 days.
Repealing Multiple Professional Practice Policies
The Board moved to repeal four Professional Practice Policies (PPPs), as part of the PODSA Modernization initiative, under the current strategic plan.
3 Professional Practice Policies recommended to be repealed; PPP-40 Repacking Bulk Nonprescription Drugs, PPP-47 Operational Procedures for Complying w/ Benzodiazepines and Other Targeted Substances Regulation, and PPP-72 Inquiry and Discipline Publication Policy. #CPBCBoard pic.twitter.com/cnQQ2VveDi
— BC Pharmacists (@BCPharmacists) June 14, 2019
The Board approved the repeal of the following PPPs, effective immediately:
PPP-40 Repackaging Bulk Non-prescription Drugs
This policy set out requirements regarding when a community pharmacy repackages bulk non-prescription drugs into smaller packages for sale.
This policy was repealed as Health Canada confirmed that the repackaging of non-prescription drugs is regulated under the Food and Drug Act and requires a Health Canada approved Drug Establishment Licence. Therefore, these repackaged drugs must be in compliance with the federal requirements, which eliminates the need for the College to also set rules on this issue.
PPP-47 Operational Procedures for Complying with Benzodiazepines and Other Targeted Substances Regulation
This policy references operational procedures for complying with the Benzodiazepines and Other Targeted Substances Regulations and the Controlled Drugs and Substances Act.
This policy was repealed as the requirements in this policy either duplicate, or are inconsistent with, federal legislation.
PPP-72 Inquiry and Discipline Publication Policy
This policy states how the College’s Inquiry and Discipline results and citations will be published pursuant to relevant provisions of the Health Professions Act.
This policy was repealed as it does not set out requirements or guidelines for registrants, which is the purpose of these professional practice policies.
Recognized Pharmacy Education Programs
The Board approved housekeeping amendments to the Health Professions Act (HPA) Bylaws Schedule C – Recognized Education Programs, for filing with the Minister of Health.
Schedule “C” under the HPA Bylaws lists pharmacy education programs in Canada and the United States as well as injection/intranasal drug administration programs that are recognized by the College.
Due to the development of new pharmacy education programs, program name changes and the discontinuation of certain programs, from time to time it is necessary to amend Schedule “C” to accurately reflect those changes. As such, housekeeping amendments to Schedule “C” were made to remove outdated content and reflect current program names.
The amendments have been filed with the Minister of Health and will come into effect 60 days after the filing date, in mid-August 2019.
Telepharmacy Licence Requirements – Removal of Schedules “C” and “E”
The Board approved amendments to the Pharmacy Operations and Drug Scheduling Act (PODSA) Bylaws to repeal Schedules “C” and “E”, and reference to physical requirements directly in the bylaws.
The PODSA Bylaws outline requirements for pharmacy licensure, including telepharmacies. Currently, these Bylaws require that new telepharmacy applications include diagrams, photos and videos confirming compliance with Schedules “C” and “E”. And, these Schedules are lists of all existing relevant physical requirements for telepharmacies throughout the College’s Bylaws and policies.
At their November 2018 meeting, the Board approved publicly posting amendments to the PODSA Bylaws to require that telepharmacy applicants demonstrate compliance with the physical requirements in the bylaws and applicable policies, instead of referencing Schedules “C” and “E”. No comments were received.
The amended bylaws will now be filed with the Minister of Health and will come into effect 60 days after the filing date, in mid-August 2019.
Committee Updates
Governance Committee
The Board approved a revision to add Board member evaluations to the responsibilities laid out in the Governance Committee’s Terms of Reference.
This change will enable the Governance Committee to better advise the Board on matters relating to Board governance.
Drug Administration Committee
The Board approved a revision to the Drug Administration Committee Terms of Reference, to reflect the name change of the “College of Registered Nurses of British Columbia” to the “British Columbia College of Nursing Professionals.”
Application Committee
The Board approved revisions to the Application Committee Terms of Reference, removing its responsibility to establish subcommittees and ad hoc working groups for Board appointment, to review, develop and administer and establish requirements for the purposes of the application process.
These requirements are already stated in legislation and are not subject to change.
Establishment of the Past Chairs Advisory Committee
The Board withdrew a motion for the establishment of a Past Chairs Advisory Committee. After considerable discussion, it was agreed to send the terms of reference back to the governance committee for further refinement before being brought back to the Board.
Establishment of the Registrar Evaluation and Succession Planning Committee
The Board approved the establishment of the Registrar Evaluation and Succession Planning Committee.
The role of this committee will be to oversee both the ongoing evaluation of the Registrar and the Registrar succession planning process.
 Share
Share


